GED Science Study Guide: Physical Science
Physical science is the study of how inanimate (nonliving) things exist and interact in nature. In this section, we’ll explore the following topics:
- Astronomy
- Chemistry
- Physics
Astronomy
Astronomy is a branch of science focusing on celestial objects (i.e., everything in space), space itself, and the universe.
We already covered a few astronomy topics by discussing the Solar System in the previous section. Here, we’ll explore everything beyond Neptune, the farthest planet from the Sun.
The Solar System is part of the Milky Way Galaxy, a slowly rotating disc containing 100–400 billion stars. That may seem impressive, but the Milky Way is only one of hundreds of billions of galaxies that make up the Universe — the entirety of space and everything in it.
Scientific advances in the 20th century helped astronomers make significant discoveries about the universe. The fact that all the galaxies are moving away from one another suggests that a Big Bang created all the matter in the universe. Why this Big Bang happened or what caused it is still a mystery. However, we do know that this Big Bang happened about 13.8 billion years ago.
Here are a few astronomy terms you should know going into the GED Science test:
- Asteroid: A lump of rock or metal floating through space.
- Black Hole: An area of intense gravity that sucks everything into it, even light.
- Comet: A ball of rocky ice. Comets make tails in the sky because heat from the Sun turns the ice into water vapor.
- Constellation: A pattern of stars in the sky. Constellations’ names date back hundreds or thousands of years.
- Meteor: An asteroid as it falls through Earth’s atmosphere. Many small meteors burn up due to the intense friction with the air.
- Meteorite: What remains of a meteor once it hits the Earth. Studying meteorites can tell scientists much about the early Solar System.
- Supernova: The implosion of an old, massive star. Supernovae are visible from tens of thousands of light-years (the distance light can travel in a year) away.
Chemistry
Chemistry is a branch of science focusing on matter (or “stuff”) and how that matter interacts.
Matter typically exists in one of three states:
- Gas (e.g., water vapor)
- Liquid (e.g., liquid water)
- Solid (e.g., ice)
The temperature at which matter turns from one state to another depends on the strength of its molecular bonds. Most metals have very high melting points because their bonds are very strong.
Chemists organize elements using the periodic table.

Source: Sandbh, CC BY-SA 4.0, via Wikimedia Commons
The periodic table organizes elements by their similarities. For example, the noble gases (Group 18) are all colorless gases that do not react with other elements.
Elements can create compounds with other elements through chemical reactions. One notable example is that, when chlorine (a poisonous gas) and sodium (a metal that explodes on contact with water) react, they produce harmless and tasty table salt.
Chemical reactions like the one below must always be balanced, meaning that both sides of the equation must have the same number of atoms.
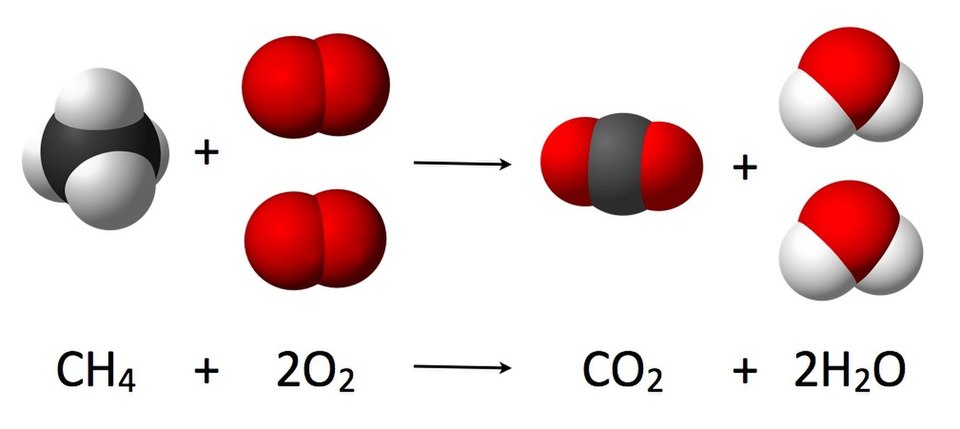
Source: JyntoRobert A. RohdeJacek FHJynto, Public domain, via Wikimedia Commons
In this equation, one methane (CH4) molecule reacts with two oxygen (O2) molecules to produce one carbon dioxide (CO2) molecule and two water (H2O) molecules.
This is a dramatic reaction, as methane and oxygen will burn in the presence of a flame. The methane and oxygen are reagents, and the carbon dioxide and water are products. The law of the conservation of matter states that chemical reactions, no matter how dramatic, cannot destroy atoms. As a result, both sides of an equation must be balanced.
Many chemistry experiments involve creating solutions, which are mixtures of two or more things. Saltwater is a simple solution. In it, the water is the solvent, or the thing something dissolves into. The salt is the solute, or the thing that dissolves into the solvent. However, in a solution, no chemical reaction takes place. If you boiled a pot of saltwater, the water would turn to steam, leaving the salt in the pan.
Physics
Physics is the branch of science that studies the underlying laws and mechanisms that show how the universe works, from the biggest stars to the smallest atoms.
To start our journey, we need to return to the humble atom.
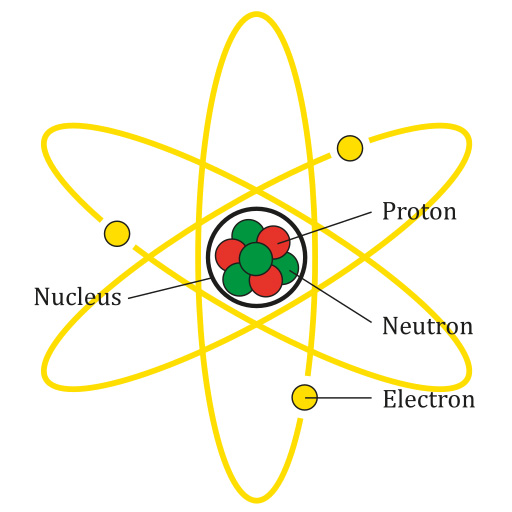
Source: AG Caesar, CC BY-SA 4.0, via Wikimedia Commons
An atom is the smallest part of any element. It contains a few important parts you should know for the GED Science test.
- Nucleus: The center of the atom containing protons and neutrons.
- Proton: A positively charged subatomic particle. An atom’s number of protons determines its atomic number on the periodic table.
- Neutron: A subatomic particle with no electric charge. Every element besides hydrogen has neutrons.
- Electron: A negatively charged subatomic particle that orbits the nucleus. Chemical reactions cause atoms to share or exchange electrons.
Two atoms that are the same element (i.e., have the same number of protons) can have different numbers of neutrons. This discovery led scientists to adopt the word isotope to describe these differences. For example, the most abundant carbon isotope in nature (like that in your pencil graphite) is carbon-12, which has 12 neutrons. However, some carbon is carbon-14, which has 14 neutrons. Because carbon-14 is somewhat unstable, it releases energy in the form of mild radiation.
Note
Radiation refers to the emitting of energy. Not all radiation is the same. You feel the warm solar radiation every time you walk outside. Some radiation is harmful because it can damage or kill our cells.
Remember in the last section when we discussed the law of the conservation of matter? There is an extra piece of information you should know. Although matter can’t be destroyed, it can be turned into energy. This is what happens in nuclear fission, where unstable atoms are split apart. The number of atoms that split is the difference between a nuclear power plant (controlled splitting) and a nuclear bomb (uncontrolled splitting).
Physical laws guide how matter interacts. Some of the oldest are Sir Isaac Newton’s (1643–1727) Laws of Motion:
- First Law: Objects will remain at rest unless acted upon by something else.
- Second Law: The rate of change is equal to the force (e.g., a push or pull) applied to something.
- Third Law: For every action, there is an equal and opposite reaction.
Physicists also study fundamental forces.
| The Four Fundamental Forces | |
| Gravity | Causes objects with greater mass to attract objects with less mass. |
| Strong nuclear force | Holds together the nucleus of each atom. Overcoming this force and splitting this atom releases energy (e.g., nuclear energy/weapons). |
| Weak nuclear force | Causes particles to decay into energy (e.g., radiation) over time. |
| Electromagnetic force | Creates forces of attraction and repulsion that keep atoms in a structure together. |
Once you have carefully reviewed this content, take the physical science review quiz below to practice your test-taking skills on questions similar to those of the official GED.